Principais vídeos


Phylum Cnidaria This phylum is famous for having stinging cells. Jellyfish and sea anemones are phylum cnidaria examples.
Phylum cnidaria characteristics include stinging cells, symmetry, and are all invertebrates.
Keywords: cnidaria biology sponge porifera 15
evolution animalia species
anemone coral polyp medusa
jellies stinging animals
classification cnidarian
You may also enjoy..
The phylum cnidaria
https://www.youtube.com/watch?v=AjM-WOeM3uA
Cnidarian classification
https://www.youtube.com/watch?v=f4h77EptlW4
-~-~~-~~~-~~-~-
Please watch: "Study Skills Teacher's Secret Guide to your Best Grades"
https://www.youtube.com/watch?v=f3bsg8gaSbw
-~-~~-~~~-~~-~-
*
*
For more Life Science videos and summaries see,
http://www.moomoomath.com/Midd....le-School-Science-an


What is Ecology the study of? Ecology is the study of how organisms interact with one another and their environment. In order to help understand what ecology is I give examples of Ecology.
These interactions can be between biotic and abiotic factors along with different levels of an organization like an organism, a community, a population or an ecosystem.
Transcript
http://www.moomoomathblog.com/....2021/03/what-is-ecol
More Information
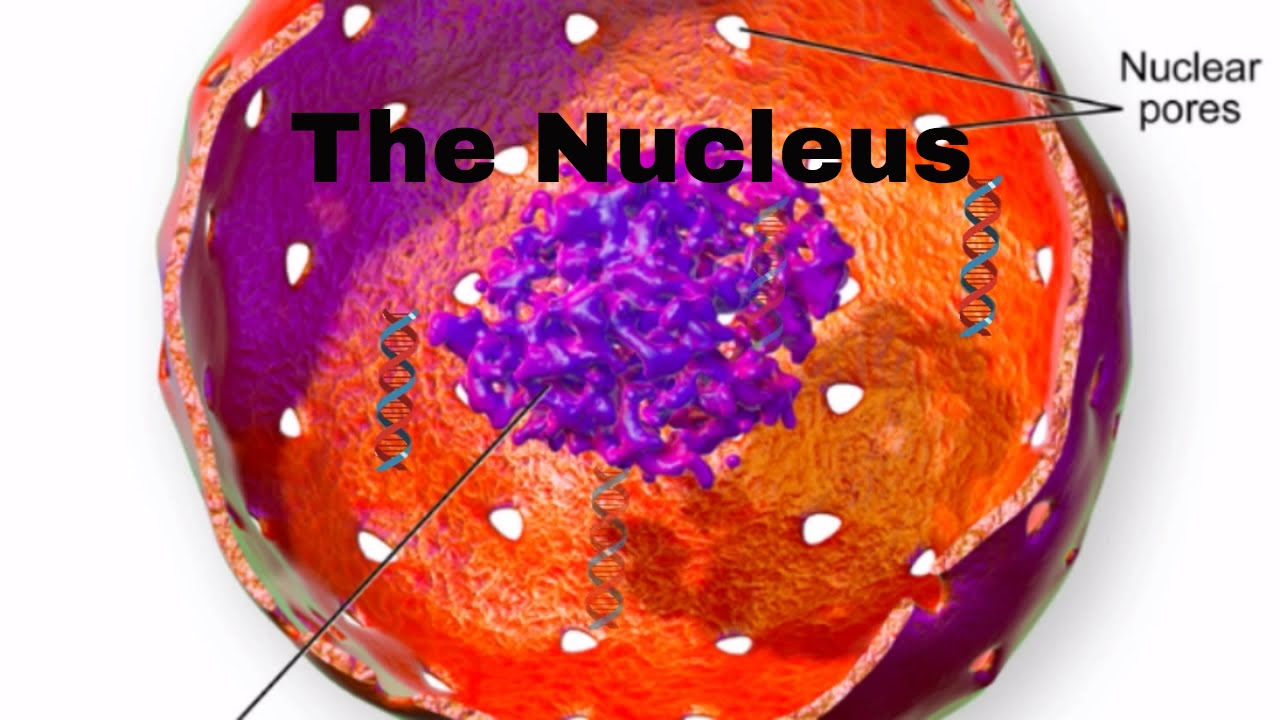

Nucleus Structure
What the parts of the nucleus?
Cell parts and function-I have always heard that the nucleus is the brain of the cell. But why is it the brain, and what are the major parts of the nucleus?
The nucleus is a large organelle. In fact, you can see the nucleus with a compound microscope.
Here it is inside a cheek cell.
I’m going to look at four main components of a nucleus and the role each part plays.
The nucleus is surrounded by a nuclear membrane. It is very similar to the cell membrane, and it is composed of two layers. It protects and houses the other parts of the nucleus.
You can also find ribosomes on the outside of the nuclear membrane.
Up next are nuclear pores. These are openings found on the nuclear membrane.
They allow items to move in and out of the nucleus. An example would be the mrna. It would travel through a nuclear pore to leave the nucleus.
Nucleoplasm This is a gel-like material found inside the nuclear membrane. You will find the DNA suspended in the nucleoplasm
Nucleolus. This is almost like a nucleus inside the nucleus. It has a very high concentration of DNA,
It is also the site of transcription which is the starting point of protein synthesis.
This is how the nucleus controls the cell. It has all the information on how to make stuff like proteins inside the cell. These proteins are used for many purposes inside the cell.
Ribosomes are also made at the nucleolus
All eukaryotic cells have a nucleus and bacteria and other prokaryotic cells do not have a nucleus.
-~-~~-~~~-~~-~-
Please watch: "Study Skills Teacher's Secret Guide to your Best Grades"
https://www.youtube.com/watch?v=f3bsg8gaSbw
-~-~~-~~~-~~-~-


Desert Biome is found on every continent. A desert is an area that receives rainfall of 10 inches or less a year.
Deserts can be very hot or very cold.
For example, the Sahara desert is the largest hot desert, and the Gobi desert in China is very large and cold most of the year.
Animals and plants in the desert must have adaptations that allow them to survive on little water.
You may enjoy my visit to a Desert Biome
https://youtu.be/jOKfQXav6Hw
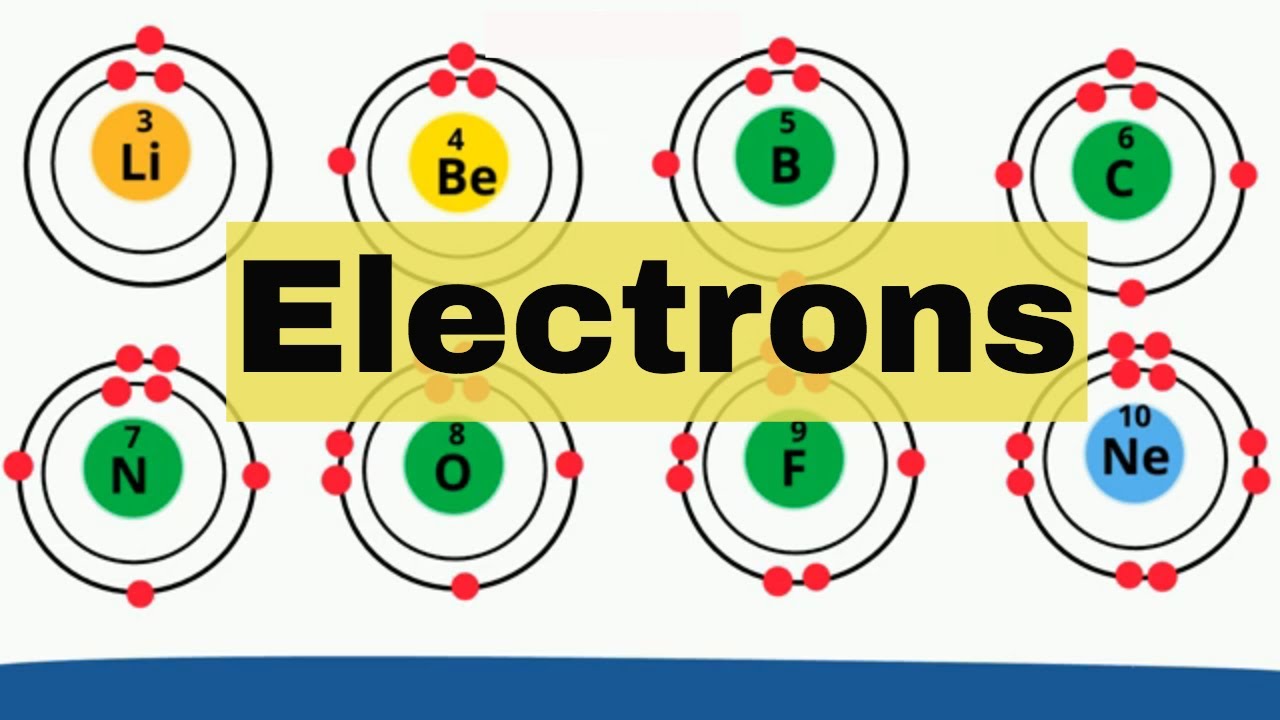

An atom is composed of a dense core called the nucleus containing protons and neutrons and a series of outer shells occupied by orbiting electrons
An electron shell may be thought of as an orbit that the electrons follow. The closest shell to the nucleus is called the "1 shell" also called "K shell", followed by the "2 shells" "L shell"), then the "3 shells" (or "M shell"), and so on.
A valence electron is an outer shell electron that is associated with an atom, and the formation of a chemical bond
Each shell can contain only a fixed number of electrons: For the first 18 elements, the valence electrons follow these rules. The first shell can hold up to two electrons, the second shell can hold up to eight electrons, the third shell can hold up to 8 The transition metals vary from these guidelines.
You can use the periodic table for the groups 1,2,13,14,15,16,17,18 in order to find the number of valence electrons in the outer shell with the exception of helium. For example, all elements in group two have 2 electrons in the outer shell.
Elements in the same period have the same number of energy shells. Period 2 will have two energy shells.
So let’s go through the elements
Up first is period 1 Element 1 is Hydrogen in group 1 and contains one energy shell and 1 electron
Element 2 Helium period 1 group 18 and is the one exception because it has 2 valence electrons in the outer shell
Now period 2 which each have 2 energy shells.
Lithium found in group 1 has 2 electrons in k shell and 1 valence electron in L
Beryllium is found in group 2 and has 2 electrons in k shell and 2 valence electrons in L
Boron Is found in group 13 and has 2 electrons in k and 3 valence electrons in L
Carbon Is found in group 14 and has 2 electrons in k and 4 valence electrons in L
Nitrogen Is found in group 15 and has 2 electrons in k and 5 valence electrons in L
Oxygen Is found in group 16 and has 2 electrons in k and 6 valence electrons in L
Fluorine Is found in group 17 and has 2 electrons in k and 7 valence electrons in L
Neon Is found in group 18 and has 2 electrons in k and 8 valence electrons in L
Now let’s move on to period 3 with 3 electron shells
Sodium Is found in group 1 and has 2 electrons in k and 8 electrons in L and 1 valence electron in m
Magnesium Is found in group 2 and has 2 electrons in k and 8 electrons in L and 2 valence electrons in m shell.
Transcript
http://www.moomoomathblog.com/....2020/01/electron-dis


Middle School Tips
Middle School Survival Guide You can survive middle school and actually enjoy it.
Here are ten tasks you should do that I have learned from 25 years of teaching middle school that will help you in these middle grades.
These tips will help you improve your grades and my straight-A students practice most of these strategies.
The 10 strategies for not only surviving middle school but having success in school
1. Use an agenda daily
2. Learn to plan ahead
3. Learn memory hacks
Here is a video that is a good starting point for memory hacks
https://www.youtube.com/watch?v=q7cPj7lSKo8
4. Pick good friends
5. Set goals
6. Always turn in your homework
7. Exercise
8. Get enough sleep
9. Read 7 Habits Highly Effective Teens
10. Be nice
More Information
http://www.moomoomathblog.com/....2020/07/middle-schoo
~-~~-~~~-~~-~-
Please watch: "Study Skills Teacher's Secret Guide to your Best Grades"
https://www.youtube.com/watch?v=f3bsg8gaSbw
-~-~~-~~~-~~-~-


Autotrophs vs Heterotrophs Producers vs Consumers
An autotroph is an organism that can self feed.The word autotroph comes from the Greek autos self and trophe nourishing.
Autotrophs are the producers in a food web.
Some common examples would include….
Autotrophs produce their energy from the sun in a chemical process called photosynthesis
They take Carbon Dioxide plus water and sunlight and produce sugar and oxygen
Autotrophs form the base of the energy pyramid.
A heterotroph comes from the greek word meaning heteros = another, different and trophe = nutrition
Heterotroph get their energy by eating other plants and animals.
They use
Sugar + oxygen to produce carbon dioxide + water and energy in the form of ATP
C6H12O6 + 6 O2 yield 6 CO2 + 6 H2O + Energy
This process is called cellular respiration
Some Heterotrophs that only eat plants, herbivores others eat only meat, carnivores others eat plants and animals omnivores while some heterotrophs feed on dead organic remains of other organisms decomposers
Updated Version: https://youtu.be/PzzCOEDtiMM
-~-~~-~~~-~~-~-
Please watch: "Study Skills Teacher's Secret Guide to your Best Grades"
https://www.youtube.com/watch?v=f3bsg8gaSbw
-~-~~-~~~-~~-~-
*
For more Life Science videos and summaries see,
http://www.moomoomath.com/Midd....le-School-Science-an
For additional information on heterotrophs:
http://www.moomoomath.com/type....s-of-heterotrophs-wi


The scientific method is a systematic method for solving problems. In this video, I review 6 steps of the scientific method.
Step 1. State the problem in a question
Step. 2 Research
Step 3. State a hypothesis
Step 4. Experiment
Step 5. State results
Step. 6 State your conclusion
Transcript
http://www.moomoomathblog.com/....2019/08/steps-of-sci
0:00 Introduction
0:04 Steps of the Sclentific Method
0:38 Step 3. Hypothesis
1:11 Independent variable is the
1:24 Dependent variable changes
1:37 Control Variables are kept constant
2:27 Step 6 Conclusion
Science Variables
http://www.moomoomathblog.com/....2019/02/learn-scienc


In this video, I cover 3 types of convergent boundaries.
Oceanic and Oceanic, Oceanic and Continental, Continental and Continental lithosphere
I also cover 2 types of divergent boundaries.
Divergent boundaries occur when plate boundaries move away from one another.
Oceanic and Oceanic plus Continental and Continental
Transform boundaries occur when plates move past one another.
More on Plate Boundaries
http://www.moomoomathblog.com/....2021/03/how-tectonic


Learn the parts of the microscope and how to focus a compound microscope.
Using a compound microscope can be fun and unlocks a whole new world.
In this video I point out a couple of tips and tricks you can use to help you use a microscope properly.
-~-~~-~~~-~~-~-
Please watch: "Study Skills Teacher's Secret Guide to your Best Grades"
https://www.youtube.com/watch?v=f3bsg8gaSbw
-~-~~-~~~-~~-~-
*
*
For more Life Science videos and summaries see,
http://www.moomoomath.com/Midd....le-School-Science-an


Learn the difference between a autotroph and a heterotroph. All plants and other organisms that produce their own food in an ecosystem are called autotrophs. Or self feeders
An Autotrophs is an organism that collects energy from sunlight or inorganic substances to produce food.
A Heterotroph is an organism that gets its energy requirement by consuming other organisms.
They are also called consumers
A heterotroph that eats only planed is called a herbivore
Heterotrophs that eat other heterotrophs are called carnivores. Lions,wolves
An organism that eats plants and animals are omnivores.
More on Autotrophs
http://www.moomoomathblog.com/....2022/05/autotrophs-a
Autotroph and Heterotroph
https://moomoomath.com/autotro....ph-vs-heterotroph-pr


Properties of the igneous rock rhyolite
Light in color there it is classified as felsic
Rhyolite contains over 70% silica which gives this rock it a light color
Because the crystals are so small it is an extrusive igneous rock and cooled quickly
in rhyolite the crystals are often too small to see. This is due to the rapid cooling of the rhyolite lava
Rhyolite is produced by violent volcanic eruptions.
Density 2.4 -2.6 g/cm3
The texture of Rhyolite is fined grain and is classified as aphanitic
In summary
Igneous
Felsic
Extrusive
Fine-grained Aphanitic
Rhyolite picture
https://www.flickr.com/photos/....jsjgeology/845670838